| Record Information |
|---|
| Version | 2.0 |
|---|
| Creation Date | 2012-05-31 14:01:53 -0600 |
|---|
| Update Date | 2015-06-03 15:54:36 -0600 |
|---|
| Secondary Accession Numbers | |
|---|
| Identification |
|---|
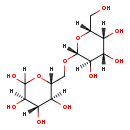
| Name: | Allolactose |
|---|
| Description | Allolactose is a disaccharide similar to lactose. It consists of the monosaccharides alpha-D-galactose and alpha-D-glucose linked through a beta 1-6 glycosidic linkage. |
|---|
| Structure | |
|---|
| Synonyms: | |
|---|
| Chemical Formula: | C12H22O11 |
|---|
| Weight: | Average: 342.2965
Monoisotopic: 342.116211546 |
|---|
| InChI Key: | DLRVVLDZNNYCBX-VDGMBKLFSA-N |
|---|
| InChI: | InChI=1S/C12H22O11/c13-1-3-5(14)8(17)10(19)12(23-3)21-2-4-6(15)7(16)9(18)11(20)22-4/h3-20H,1-2H2/t3-,4-,5+,6-,7+,8+,9-,10-,11?,12-/m1/s1 |
|---|
| CAS number: | 645-03-4 |
|---|
| IUPAC Name: | (3R,4S,5S,6R)-6-({[(2R,3R,4S,5R,6R)-3,4,5-trihydroxy-6-(hydroxymethyl)oxan-2-yl]oxy}methyl)oxane-2,3,4,5-tetrol |
|---|
| Traditional IUPAC Name: | allolactose |
|---|
| SMILES: | [H]C1(O)O[C@]([H])(CO[C@]2([H])O[C@]([H])(CO)[C@]([H])(O)[C@]([H])(O)[C@@]2([H])O)[C@@]([H])(O)[C@]([H])(O)[C@@]1([H])O |
|---|
| Chemical Taxonomy |
|---|
| Description | belongs to the class of organic compounds known as o-glycosyl compounds. These are glycoside in which a sugar group is bonded through one carbon to another group via a O-glycosidic bond. |
|---|
| Kingdom | Organic compounds |
|---|
| Super Class | Organic oxygen compounds |
|---|
| Class | Organooxygen compounds |
|---|
| Sub Class | Carbohydrates and carbohydrate conjugates |
|---|
| Direct Parent | O-glycosyl compounds |
|---|
| Alternative Parents | |
|---|
| Substituents | - O-glycosyl compound
- Disaccharide
- Oxane
- Secondary alcohol
- Hemiacetal
- Oxacycle
- Organoheterocyclic compound
- Polyol
- Acetal
- Hydrocarbon derivative
- Primary alcohol
- Alcohol
- Aliphatic heteromonocyclic compound
|
|---|
| Molecular Framework | Aliphatic heteromonocyclic compounds |
|---|
| External Descriptors | |
|---|
| Physical Properties |
|---|
| State: | Not Available |
|---|
| Charge: | 0 |
|---|
| Melting point: | Not Available |
|---|
| Experimental Properties: | |
|---|
| Predicted Properties | |
|---|
| Biological Properties |
|---|
| Cellular Locations: | Cytoplasm |
|---|
| Reactions: | |
|---|
| SMPDB Pathways: | Not Available |
|---|
| KEGG Pathways: | Not Available |
|---|
| EcoCyc Pathways: | Not Available |
|---|
| Concentrations |
|---|
| Not Available |
|---|
| Spectra |
|---|
| Spectra: | |
|---|
| References |
|---|
| References: | - Keseler, I. M., Collado-Vides, J., Santos-Zavaleta, A., Peralta-Gil, M., Gama-Castro, S., Muniz-Rascado, L., Bonavides-Martinez, C., Paley, S., Krummenacker, M., Altman, T., Kaipa, P., Spaulding, A., Pacheco, J., Latendresse, M., Fulcher, C., Sarker, M., Shearer, A. G., Mackie, A., Paulsen, I., Gunsalus, R. P., Karp, P. D. (2011). "EcoCyc: a comprehensive database of Escherichia coli biology." Nucleic Acids Res 39:D583-D590. Pubmed: 21097882
- van der Werf, M. J., Overkamp, K. M., Muilwijk, B., Coulier, L., Hankemeier, T. (2007). "Microbial metabolomics: toward a platform with full metabolome coverage." Anal Biochem 370:17-25. Pubmed: 17765195
- Winder, C. L., Dunn, W. B., Schuler, S., Broadhurst, D., Jarvis, R., Stephens, G. M., Goodacre, R. (2008). "Global metabolic profiling of Escherichia coli cultures: an evaluation of methods for quenching and extraction of intracellular metabolites." Anal Chem 80:2939-2948. Pubmed: 18331064
|
|---|
| Synthesis Reference: | Not Available |
|---|
| Material Safety Data Sheet (MSDS) | Not Available |
|---|
| Links |
|---|
| External Links: | |
|---|