| Record Information |
|---|
| Version | 2.0 |
|---|
| Creation Date | 2012-05-31 13:56:04 -0600 |
|---|
| Update Date | 2015-06-03 15:54:15 -0600 |
|---|
| Secondary Accession Numbers | |
|---|
| Identification |
|---|
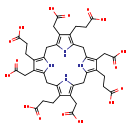
| Name: | Uroporphyrinogen I |
|---|
| Description | Uroporphyrinogens are porphyrinogen variants in which each pyrrole ring has one acetate side chain and one propionate side chain; it is formed by condensation 4 four molecules of porphobilinogen. 4 isomers are possible but only 2 commoly are found, types and I. Uroporphyrinogen I is a functional intermediate in heme biosynthesis while Uroporphyrinogen is produced in an abortive side reaction. |
|---|
| Structure | |
|---|
| Synonyms: | - 3,8,13,18-Tetrakis(carboxymethyl)-5,10,15,20,22,24-hexahydro-(8CI)-2,7,12,17-Porphinetetrapropionate
- 3,8,13,18-Tetrakis(carboxymethyl)-5,10,15,20,22,24-hexahydro-(8CI)-2,7,12,17-Porphinetetrapropionic acid
- 3,8,13,18-Tetrakis(carboxymethyl)-5,10,15,20,22,24-hexahydro-21H,23H-Porphine-2,7,12,17-tetrapropanoate
- 3,8,13,18-Tetrakis(carboxymethyl)-5,10,15,20,22,24-hexahydro-21H,23H-Porphine-2,7,12,17-tetrapropanoic acid
- 3,8,13,18-Tetrakis(carboxymethyl)-5,10,15,20,22,24-hexahydroporphyrin-2,7,12,17-tetrapropanoate
- 3,8,13,18-Tetrakis(carboxymethyl)-5,10,15,20,22,24-hexahydroporphyrin-2,7,12,17-tetrapropanoic acid
- Uroporphyrinogen I
- Uroporphyrinogen III
- Uroporphyrinogen-I
|
|---|
| Chemical Formula: | C40H44N4O16 |
|---|
| Weight: | Average: 836.7946
Monoisotopic: 836.27523138 |
|---|
| InChI Key: | QTTNOSKSLATGQB-UHFFFAOYSA-N |
|---|
| InChI: | InChI=1S/C40H44N4O16/c45-33(46)5-1-17-21(9-37(53)54)29-14-26-19(3-7-35(49)50)23(11-39(57)58)31(43-26)16-28-20(4-8-36(51)52)24(12-40(59)60)32(44-28)15-27-18(2-6-34(47)48)22(10-38(55)56)30(42-27)13-25(17)41-29/h41-44H,1-16H2,(H,45,46)(H,47,48)(H,49,50)(H,51,52)(H,53,54)(H,55,56)(H,57,58)(H,59,60) |
|---|
| CAS number: | 1867-62-5 |
|---|
| IUPAC Name: | 3-[9,14,19-tris(2-carboxyethyl)-5,10,15,20-tetrakis(carboxymethyl)-21,22,23,24-tetraazapentacyclo[16.2.1.1³,⁶.1⁸,¹¹.1¹³,¹⁶]tetracosa-1(20),3,5,8,10,13,15,18-octaen-4-yl]propanoic acid |
|---|
| Traditional IUPAC Name: | uroporphyrinogen I |
|---|
| SMILES: | OC(=O)CCC1=C2CC3=C(CC(O)=O)C(CCC(O)=O)=C(CC4=C(CC(O)=O)C(CCC(O)=O)=C(CC5=C(CC(O)=O)C(CCC(O)=O)=C(CC(N2)=C1CC(O)=O)N5)N4)N3 |
|---|
| Chemical Taxonomy |
|---|
| Description | belongs to the class of organic compounds known as porphyrins. Porphyrins are compounds containing a fundamental skeleton of four pyrrole nuclei united through the alpha-positions by four methine groups to form a macrocyclic structure. |
|---|
| Kingdom | Organic compounds |
|---|
| Super Class | Organoheterocyclic compounds |
|---|
| Class | Tetrapyrroles and derivatives |
|---|
| Sub Class | Porphyrins |
|---|
| Direct Parent | Porphyrins |
|---|
| Alternative Parents | |
|---|
| Substituents | - Porphyrin
- Substituted pyrrole
- Pyrrole
- Heteroaromatic compound
- Carboxylic acid derivative
- Carboxylic acid
- Azacycle
- Carbonyl group
- Hydrocarbon derivative
- Organic oxide
- Organopnictogen compound
- Organooxygen compound
- Organonitrogen compound
- Organic oxygen compound
- Organic nitrogen compound
- Aromatic heteropolycyclic compound
|
|---|
| Molecular Framework | Aromatic heteropolycyclic compounds |
|---|
| External Descriptors | |
|---|
| Physical Properties |
|---|
| State: | Solid |
|---|
| Charge: | -8 |
|---|
| Melting point: | Not Available |
|---|
| Experimental Properties: | |
|---|
| Predicted Properties | |
|---|
| Biological Properties |
|---|
| Cellular Locations: | Cytoplasm |
|---|
| Reactions: | |
|---|
| SMPDB Pathways: | |
|---|
| KEGG Pathways: | - Porphyrin and chlorophyll metabolism ec00860
|
|---|
| EcoCyc Pathways: | |
|---|
| Concentrations |
|---|
| Not Available |
|---|
| Spectra |
|---|
| Spectra: | | Spectrum Type | Description | Splash Key | |
|---|
| Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Positive | splash10-0gi0-0000000790-66d58d00c9ac805a86dd | View in MoNA |
|---|
| Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Positive | splash10-05fr-0000000910-4ff4ea54a281f8d6b0d1 | View in MoNA |
|---|
| Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Positive | splash10-0adi-0000000900-919f095ae5cb2c3e6cd2 | View in MoNA |
|---|
| Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Negative | splash10-000i-0000000790-5f547e23817fab228be1 | View in MoNA |
|---|
| Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Negative | splash10-00y0-1000000940-4d4fc3a395b4bfdd41db | View in MoNA |
|---|
| Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Negative | splash10-05fr-5000000900-f49d69c9724b023caf67 | View in MoNA |
|---|
| Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Positive | splash10-0g4i-0000000960-0125e055eb61d33157a3 | View in MoNA |
|---|
| Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Positive | splash10-000t-0000000910-6066cce322481af52283 | View in MoNA |
|---|
| Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Positive | splash10-000t-0000000900-a0d3dd169d62d3b9101a | View in MoNA |
|---|
| Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Negative | splash10-0002-0000000900-e9ab543d3cd6074dec5d | View in MoNA |
|---|
| Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Negative | splash10-0002-0000000900-d45cf1d17beafd43554e | View in MoNA |
|---|
| Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Negative | splash10-00kb-0000000900-b910cfcdb3042465a8f1 | View in MoNA |
|---|
| MS | Mass Spectrum (Electron Ionization) | Not Available | View in JSpectraViewer |
|---|
| 1D NMR | 13C NMR Spectrum | Not Available | View in JSpectraViewer |
|---|
| 1D NMR | 1H NMR Spectrum | Not Available | View in JSpectraViewer |
|---|
| 1D NMR | 13C NMR Spectrum | Not Available | View in JSpectraViewer |
|---|
| 1D NMR | 1H NMR Spectrum | Not Available | View in JSpectraViewer |
|---|
| 1D NMR | 13C NMR Spectrum | Not Available | View in JSpectraViewer |
|---|
| 1D NMR | 1H NMR Spectrum | Not Available | View in JSpectraViewer |
|---|
| 1D NMR | 13C NMR Spectrum | Not Available | View in JSpectraViewer |
|---|
| 1D NMR | 1H NMR Spectrum | Not Available | View in JSpectraViewer |
|---|
| 1D NMR | 13C NMR Spectrum | Not Available | View in JSpectraViewer |
|---|
| 1D NMR | 1H NMR Spectrum | Not Available | View in JSpectraViewer |
|---|
| 1D NMR | 13C NMR Spectrum | Not Available | View in JSpectraViewer |
|---|
| 1D NMR | 1H NMR Spectrum | Not Available | View in JSpectraViewer |
|---|
| 1D NMR | 13C NMR Spectrum | Not Available | View in JSpectraViewer |
|---|
| 1D NMR | 1H NMR Spectrum | Not Available | View in JSpectraViewer |
|---|
| 1D NMR | 13C NMR Spectrum | Not Available | View in JSpectraViewer |
|---|
| 1D NMR | 1H NMR Spectrum | Not Available | View in JSpectraViewer |
|---|
| 1D NMR | 13C NMR Spectrum | Not Available | View in JSpectraViewer |
|---|
| 1D NMR | 1H NMR Spectrum | Not Available | View in JSpectraViewer |
|---|
| 1D NMR | 13C NMR Spectrum | Not Available | View in JSpectraViewer |
|---|
| 1D NMR | 1H NMR Spectrum | Not Available | View in JSpectraViewer |
|---|
|
|---|
| References |
|---|
| References: | - Kanehisa, M., Goto, S., Sato, Y., Furumichi, M., Tanabe, M. (2012). "KEGG for integration and interpretation of large-scale molecular data sets." Nucleic Acids Res 40:D109-D114. Pubmed: 22080510
- Keseler, I. M., Collado-Vides, J., Santos-Zavaleta, A., Peralta-Gil, M., Gama-Castro, S., Muniz-Rascado, L., Bonavides-Martinez, C., Paley, S., Krummenacker, M., Altman, T., Kaipa, P., Spaulding, A., Pacheco, J., Latendresse, M., Fulcher, C., Sarker, M., Shearer, A. G., Mackie, A., Paulsen, I., Gunsalus, R. P., Karp, P. D. (2011). "EcoCyc: a comprehensive database of Escherichia coli biology." Nucleic Acids Res 39:D583-D590. Pubmed: 21097882
- Maines MD, Mayer RD: Inhibition of testicular cytochrome P-450-dependent steroid biosynthesis by cis-platinum. Reversal by human chorionic gonadotropin. J Biol Chem. 1985 May 25;260(10):6063-8. Pubmed: 4039724
- Mukerji SK, Pimstone NR: Defective human erythrocyte uroporphyrinogen decarboxylase in familial porphyria cutanea tarda: the metabolic lesion or the result of endogenous porphyrinemia? Biochem Biophys Res Commun. 1988 Jul 15;154(1):39-46. Pubmed: 3395340
|
|---|
| Synthesis Reference: | Burton, Gerardo; Fagerness, Paul E.; Hosozawa, Shigeki; Jordan, Peter M.; Scott, A. Ian. Carbon-13 NMR evidence for a new intermediate, pre-uroporphyrinogen, in the enzymic transformation of porphobilinogen into uroporphyrinogens I and III. Journal of |
|---|
| Material Safety Data Sheet (MSDS) | Not Available |
|---|
| Links |
|---|
| External Links: | |
|---|